 
When it melts, the bulb lights up showing a current flow, and bubbles of brown gas are seen at the positive electrode (the anode). Nothing happens at all until the lead(II) bromide is molten. An experimental set-up would look something like this: One of the common compounds to electrolyse molten is lead(II) bromide - partly because the products are easily visible, and partly because its melting point isn't too high. We are starting with the simpler case - where the electrolyte is molten. Electrolysis can't happen unless the ions can move to the electrodes. It has to be one or the other because otherwise the ions can't move. 
What gets discharged at the electrodes during electrolysis depends on whether the electrolyte is molten or in solution. Negative ions, attracted to the positive anode, are known as anions. Remember PANC - positive anode, negative cathode.īecause positive ions are attracted to the negative cathode, they are known as cations. The positive electrode is called the anode the negative one the cathode. 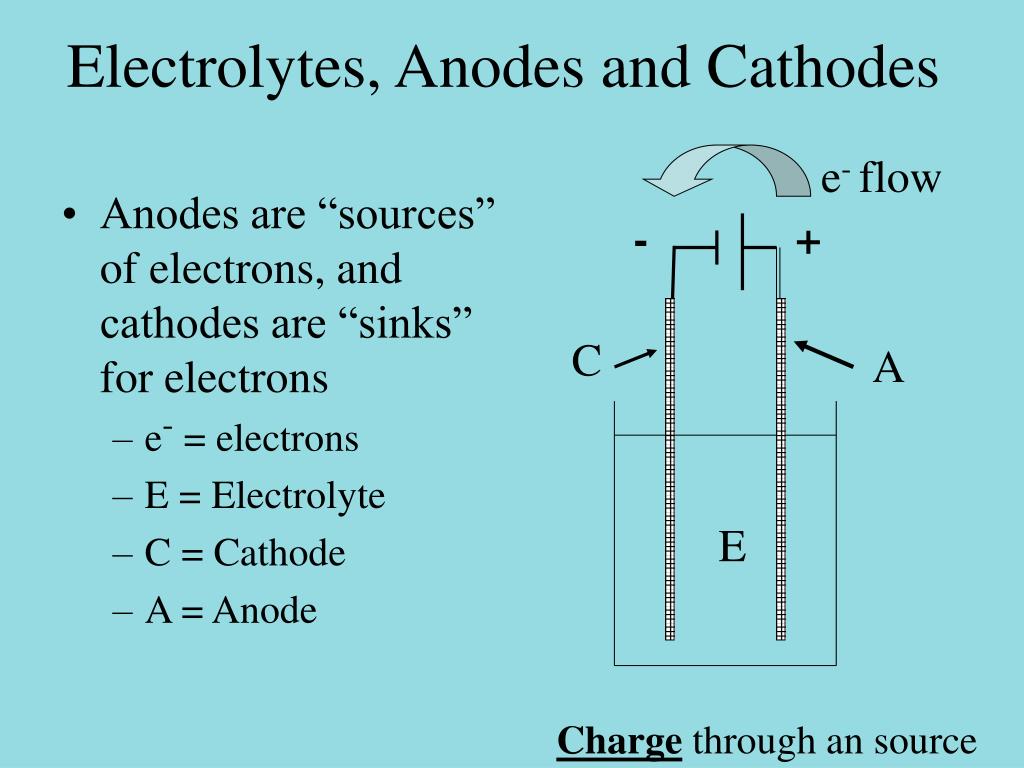
The electrodes are the pieces of carbon or metal which are placed in the electrolyte and connected to a dc power source. It also follows, of course, that an electrolyte must contain ions. The reason that an electrolyte has to be molten or in solution is that the current is carried through the electrolyte by the movement of ions - not electrons. The metal or carbon isn't chemically changed in the process.Įlectrolysis is defined as a chemical change produced when electricity is passed through a substance which is either molten or in solution.Īn electrolyte is a compound which undergoes electrolysis. The flow of electricity through a metal or carbon is due to the movement of electrons. Most people will have met all this in chemistry courses for 14 - 16 year olds.Ĭonduction of electricity through metals and carbon This page introduces the basic terms and ideas in electrolysis by looking at the electrolysis of molten compounds. Introducing electrolysis via the electrolysis of molten compounds 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
